Aminoglycosides are a group of potent antibiotics primarily used to treat certain infections caused by aerobic, Gram-negative bacteria. They are used in the treatment of severe infections of the abdomen, urinary tract, skin and soft tissue, bone, cervix, blood, eye, ear, lungs, and heart. The gram-negative bacteria include the model organism Escherichia coli and many pathogenic bacteria, such as Pseudomonas aeruginosa.
The global market for Aminoglycosides, estimated at USD 1.07 billion in 2022, is projected to reach a revised size of US$ 1.2 billion by 2028, growing at a CAGR of 1.7% from 2022-2028. The growth is due to the increasing prevalence of bacterial infections and the rising demand for effective treatments. The increasing number of regulatory approvals for new aminoglycoside drugs and the increasing adoption of these drugs by healthcare providers also drive this growth.
One major opportunity in the aminoglycosides market is the development of novel aminoglycosides with improved efficacy and reduced toxicity. Traditional aminoglycosides have been associated with side effects such as ototoxicity and nephrotoxicity, which can limit their use in certain patient populations. However, new drugs such as gentamicin liposome inhalation suspension have been developed to target specific bacterial infections in the respiratory tract, reducing the risk of systemic side effects.
Another opportunity in the aminoglycosides market is the increasing use of these drugs in combination therapies. Aminoglycosides are often used in combination with other antibiotics to enhance their effectiveness and reduce the development of antibiotic resistance. For example, amikacin is commonly used in combination with beta-lactams to treat serious infections caused by gram-negative bacteria.
The rising prevalence of antibiotic resistance is also a major driver of growth in the aminoglycosides market. As bacteria become more resistant to traditional antibiotics, the need for effective treatments increases, leading to increased demand for aminoglycosides. This trend is expected to continue as the number of antibiotic-resistant infections continues to rise.
Reports Suggested for Aminoglycosides Marketers/Manufacturers
Global key companies of Aminoglycosides Antibiotics Drug include Jemincare, Novartis, Teva, Fangyuan-pharma, Aike Pharmaceutical, etc.
Neomycin, Tobramycin, Gentamicin, Amikacin, and Etimicin mainly segment the aminoglycosides antibiotics market.
For example, Tobramycin antibiotic derived from Streptomyces tenebrarius used to treat various bacterial infections, particularly Gram-negative infections.
TOBI® PODHALER® capsule is a prescription inhaled medication for cystic fibrosis patients whose lungs contain bacteria called Pseudomonas aeruginosa. The brand is owned by Viatris, which was formed by merging Mylan N.V. with Upjohn, Pfizer's off-patent medicine division.
AGP Limited (AGP) is one of the largest pharmaceutical companies in Pakistan, and manufactures NEBCIN. Based on tobramycin sulfate, a water-soluble antibiotic of the aminoglycoside group, is derived from the actinomycete Streptomyces tenebrarius. The nebcin injection is a clear and colorless sterile aqueous solution for parenteral administration.
Novartis Pharma AG manufactures Tobrex® Ophthalmic Solution containing tobramycin, a water-soluble aminoglycoside antibiotic active against various gram-negative and gram-positive ophthalmic pathogens.
Reports Suggested for Aminoglycosides Market Investors
Tobramycin Sulfate is the sulfate salt of tobramycin. The global key manufacturers of Tobramycin Sulfate include Santa Cruz Biotechnology, AG Scientific, BioVision, LifeSpan BioSciences, Thermo Fisher Scientific, LGC, Adooq Bioscience, Boston BioProducts, and Biorbyt, etc.
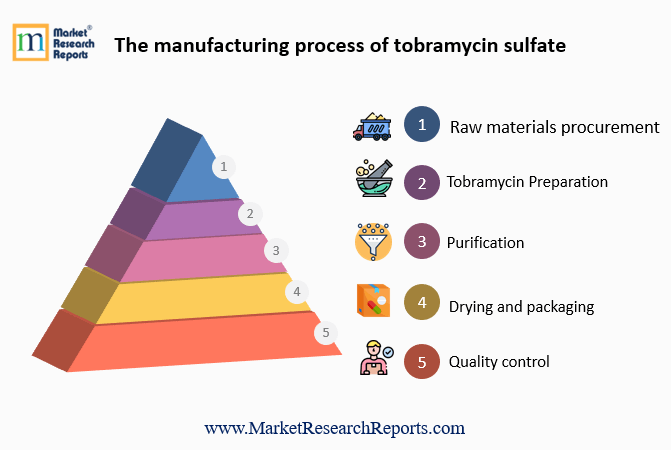
The manufacturing process of tobramycin sulfate involves several steps:
- Raw materials procurement: The first step in the manufacturing process is procuring the raw materials, which include tobramycin, sulfuric acid, and water.
- Tobramycin preparation: Tobramycin is dissolved in water to form a solution, which is then treated with sulfuric acid to convert it into its sulfate salt form.
- Purification: The resulting solution is then purified to remove any impurities and unwanted by-products. This can be done using various techniques, such as filtration, centrifugation, and crystallization.
- Drying and packaging: The purified solution is then dried to form a solid powder, which is then packaged into appropriate containers for storage and distribution.
- Quality control: Throughout the manufacturing process, various quality control measures are taken to ensure the purity and potency of the final product. These include testing the raw materials, intermediate products, and the final product for various parameters, such as potency, sterility, and stability.
- Overall, the manufacturing process of tobramycin sulfate involves the conversion of the antibiotic into its salt form, purification, drying, and packaging, with strict quality control measures to ensure the safety and effectiveness of the final product.
Reports Suggested for Chemical/Salt/API Manufacturers
Information Source: Manufacturer website and press releases
Disclaimer: The products mentioned here are not intended to diagnose, treat, cure, or prevent any disease. The information provided on this blog is marketing material for the informational purposes of pharma manufacturers, investors, and chemical manufacturers and should not be considered medical advice. Always consult with a qualified healthcare professional before taking any medication or supplement.
Related Blog Posts
- Cephalosporin Antibiotic Market: Opportunities and Challenges
- Clindamycin Antibiotic Market: Opportunities and Challenges
- Macrolide Antibiotic Market: Opportunities and Challenges