World's Top Selling Pharmaceutical Drugs and Manufacturers by Sales

As per the latest data compiled, the top selling pharmaceutical drugs are HUMIRA® (sales USD 19.93 biilion) by AbbVie Inc., REVLIMID® (sales USD 9.68 biilion) by Celgene and KEYTRUDA® (sales USD 7.17 biilion) by Merck & Co., Inc..
1 . 1.AbbVie Inc.
AbbVie's portfolio of products includes a diversified therapy that address some of the world's most complex and serious diseases. AbbVie's top 3 products for FY 2018 are HUMIRA®, IMBRUVICA® and MAVYRET™; contributes together USD 26.964 billion which is 82.33% of total AbbVie's revenue for FY 2018.

HUMIRA® (adalimumab) is a biologic therapy administered as a subcutaneous injection. It is used to treat the autoimmune diseases such as Adult Crohn's disease, Ankylosing spondylitis, Rheumatoid arthritis, Psoriatic arthritis, Axial spondyloarthropathy and others. Currently It is approved in the United States, Canada and Mexico (collectively, North America) and in the European Union. HUMIRA® is also approved in Japan for the treatment of intestinal Behçet's disease. FY 2018 revenue from HUMIRA® was USD 19.93 billion.
MAVYRET™ (glecaprevir and pibrentasvir) tablets are a prescription medicine used to treat adults with chronic (lasting a long time) hepatitis C virus (hep C) genotypes 1, 2, 3, 4, 5, or 6 infection without cirrhosis or with compensated cirrhosis. Global MAVYRET sales increased by more than 100% in 2018 as a result of market share gains following the FDA and EMA approvals of MAVYRET in the second half of 2017 as well as further geographic expansion in 2018. FY 2018 revenue from MAVYRET™ was USD 3.59 billion.
IMBRUVICA® (ibrutinib) is an oral, once-daily therapy that inhibits a protein called Bruton's tyrosine kinase (BTK). IMBRUVICA® was one of the first medicines to receive a United States Food and Drug Administration (FDA) approval after being granted a Breakthrough Therapy Designation and is one of the few therapies to receive four separate designations. IMBRUVICA® currently is approved for the treatment of adult patients with diseases such as Chronic lymphocytic leukemia, Mantle cell lymphoma, Waldenström’s macroglobulinemia, Marginal zone lymphoma (MZL) and Chronic graft versus host disease (cGVHD). FY 2018 worldwide revenue from IMBRUVICA® was USD 3.43 billion.
2 . 2.Roche Holding AG:
Roche’s sales in the Pharmaceuticals Division increased 7% to USD 43.12 billion; new medicines contributed almost all growth. Roche’s top 3 products for FY 2018 are Herceptin®, Avastin® and RITUXAN®/MabThera®; contributes together USD 18.642 billion which is 31.98% of total Roche’s revenue for FY 2018.
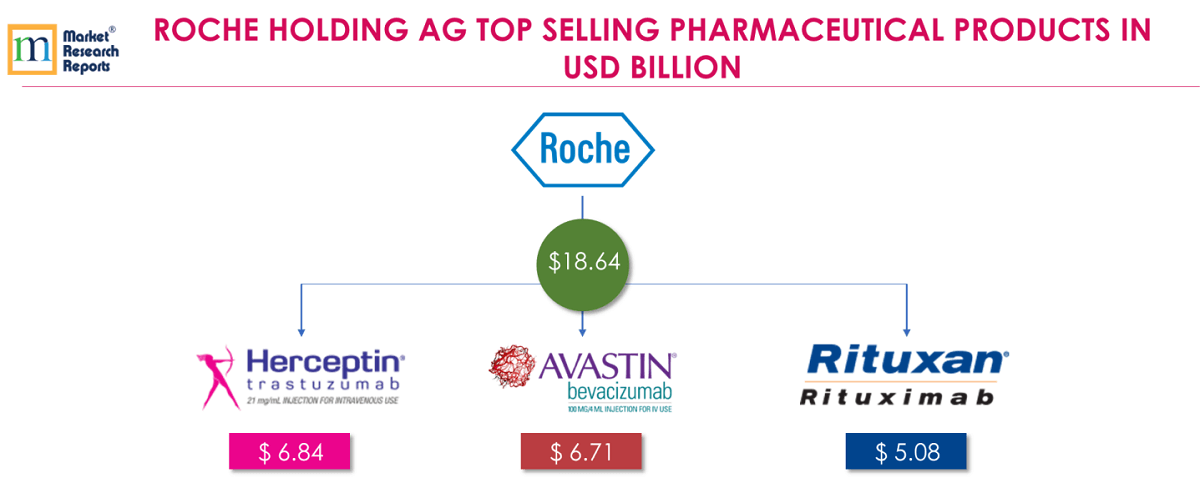
Herceptin® (Trastuzumab) is the most popular breast cancer medicine that has helped millions of women around the world. Herceptin is approved for the treatment of early-stage breast cancer that is Human Epidermal growth factor Receptor 2-positive (HER2+). Herceptin® is also approved for the treatment of HER2-positive metastatic cancer of the stomach or gastroesophageal junction. Herceptin sales increases (+1%) were mainly driven by growth in the US and in China. In Europe (-16%) and Japan (-16%), sales were affected by biosimilars. FY 2018 revenue from Herceptin® was USD 6.84 billion.
Avastin® (Bevacizumab) is approved for advanced colorectal, breast, lung, kidney, cervical and ovarian cancer and relapsed glioblastoma (a type of brain tumour). Sales increased in the International region (+12%), in the US (+1%) and in Japan (+3%) but declined in Europe (-1%). FY 2018 revenue from Avastin® was USD 6.71 billion.
RITUXAN®/MabThera® (Rituximab) is approved for forms of blood cancer, rheumatoid arthritis and certain types of vasculitis. Sales development was impacted in Europe (-47%) and Japan (-36%) by biosimilar competition. In the Europe, Asia Pacific and other parts of world Rituxan is marketed as MabThera. While in the United States, Japan and Canada MabThera is marketed as Rituxan. FY 2018 revenue from RITUXAN®/MabThera® was USD 5.08 billion.
3 . 3.Merck & Co., Inc:

KEYTRUDA® (pembrolizumab), Merck's Oncology division's anti-PD-1 (programmed death receptor-1) therapy, as monotherapy for the treatment of certain patients with non-small-cell lung cancer (NSCLC), melanoma, classical Hodgkin Lymphoma (cHL), urothelial carcinoma, head and neck squamous cell carcinoma (HNSCC), gastric orgastroesophageal junction adenocarcinoma, and microsatellite instability-high (MSI-H) or mismatch repair deficient cancer, and in combination with chemotherapy in certain patients with NSCLC. FY 2018 revenue from KEYTRUDA® was USD 7.17 billion.
JANUVIA® (sitagliptin) and Janumet (sitagliptin/metformin HCl) for the treatment of type 2 diabetes. Worldwide combined sales of Januvia and Janumet, medicines that help lower blood sugar levels in adults with type 2 diabetes, were USD 5.9 billion in 2018, essentially flat compared with 2017.
GARDASIL®9 (Human Papillomavirus 9-valent Vaccine, Recombinant) helps protect girls and women ages 9 to 26 against cervical, vaginal, vulvar, and anal cancers and genital warts caused by 9 types of HPV (human papillomavirus). FY 2018 revenue from GARDASIL®9 was USD 3.15 billion.
4 . 4.Bristol-Myers Squibb Company (BMS):
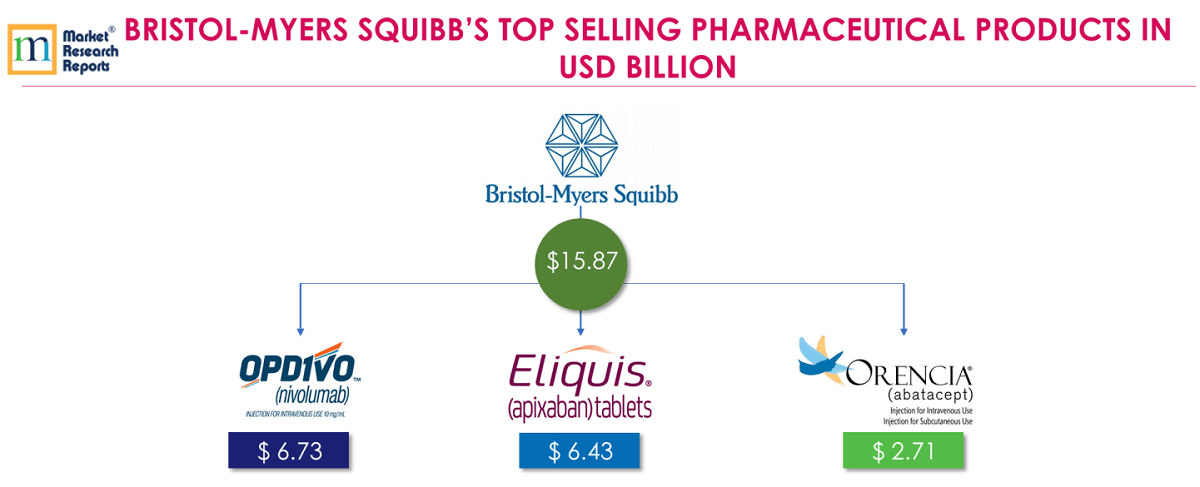
OPDIVO® (nivolumab), a biological product, is a fully human monoclonal antibody that binds to the PD-1 on T and NKT cells. OPDIVO® is a prescription medicine used to treat people with a type of advanced stage lung cancer (called non-small cell lung cancer). OPDIVO® is also approved for several anti-cancer indications including bladder, blood, colon, head and neck, kidney, liver, lung, melanoma and stomach. The Opdivo + Yervoy regimen also is approved in multiple markets for the treatment of melanoma, RCC, and CRC. FY 2018 revenue from OPDIVO® was USD 6.73 billion.
ELIQUIS® (apixaban) is an oral Factor Xa inhibitor (a class of anticoagulant drugs), targeted at stroke prevention in adult patients with NVAF and the prevention and treatment of VTE disorders. ELIQUIS® is jointly developed and is commercialized by Pfizer and BMS. FY 2018 revenue from ELIQUIS® was USD 6.43 billion.
ORENCIA® (abatacept) is a prescription medicine that reduces signs and symptoms in adults with moderate to severe RA (Rheumatoid Arthritis) and Juvenile idiopathic arthritis (JIA). BMS has got approval in Japan for an intravenously administered treatment of moderate to severe polyarticular JIA in patients two years of age and older in Feb 2018. FY 2018 revenue from ORENCIA® was USD 2.71 billion.
5 . 5.Pfizer Inc:
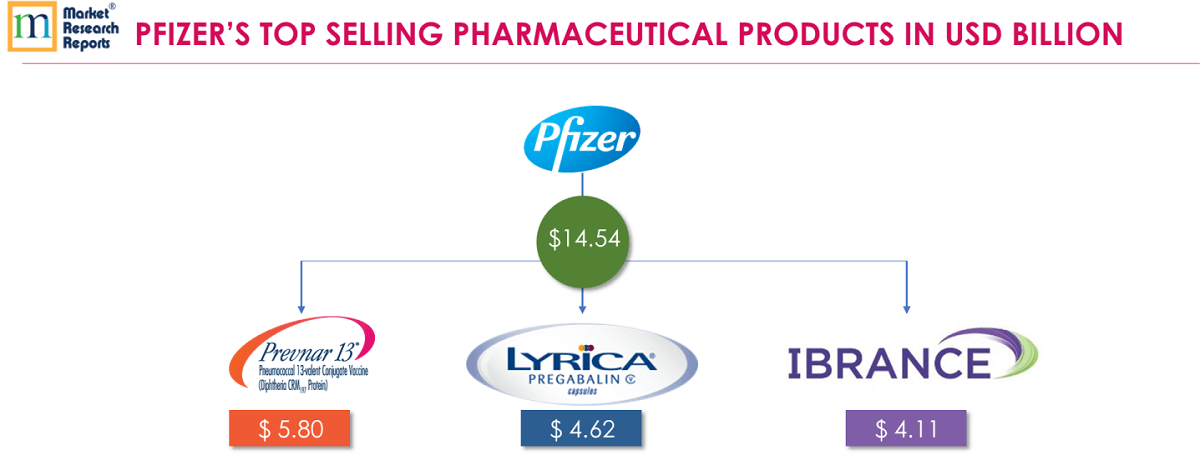
PREVNAR 13® (Pneumococcal 13-valent conjugate vaccine [diphtheria CRM197 protein]) is a vaccine indicated in children 6 weeks through 17 years for active immunization for the prevention of invasive disease caused by Streptococcus pneumoniae serotypes. In adults 18 years of age and older, Prevnar 13® is indicated for active immunization for the prevention of pneumonia and invasive disease caused by Streptococcus pneumoniae serotypes in the vaccine. FY 2018 revenue from PREVNAR 13® was USD 5.80 billion.
LYRICA® (pregabalin) is a prescription medicine approved by the U.S. Food and Drug Administration (FDA) to treat fibromyalgia, diabetic nerve pain, spinal cord injury nerve pain and pain after shingles. LYRICA® is also indicated to treat partial onset seizures in adults with epilepsy. The operational growth in worldwide LYRICA® revenues in Innovative Health segment in 2018 was primarily due to growth in the U.S. and growth in the orally dissolving tablet formulation in Japan, partially offset by losses of exclusivity primarily in Australia. FY 2018 revenue from LYRICA® was USD 4.62 billion.
IBRANCE® (palbociclib) is a targeted therapy known as a CDK 4/6 inhibitor. IBRANCE® medication is approved for women with hormone receptor positive (HR+), human epidermal growth factor receptor 2-negative (HER2-) metastatic breast cancer—also known as HR+/HER2-. IBRANCE® maintains class leadership among cyclin-dependent kinase inhibitors in major markets, supported by Pfizer's scientific/clinical data and continued positive patient experience. FY 2018 revenue from IBRANCE® was USD 4.118 billion.
6 . 6.Johnson & Johnson:
Johnson & Johnson's Pharmaceutical segment is focused on six therapeutic areas: Immunology (e.g., rheumatoid arthritis, inflammatory bowel disease and psoriasis), Infectious Diseases and Vaccines (e.g., HIV/AIDS), Neuroscience (e.g., mood disorders, neurodegenerative disorders and schizophreni ), Oncology (e.g., prostate cancer and hematologic malignancies), Cardiovascular and Metabolism (e.g., thrombosis and diabetes) and Pulmonary Hypertension (e.g.,Pulmonary Arterial Hypertension). Johnson & Johnson's top 3 products for FY 2018 are REMICADE®, STELARA® and ZYTIGA®; contributes together USD 13.98 billion which is 34.32% of total pharmaceutical revenue for FY 2018.

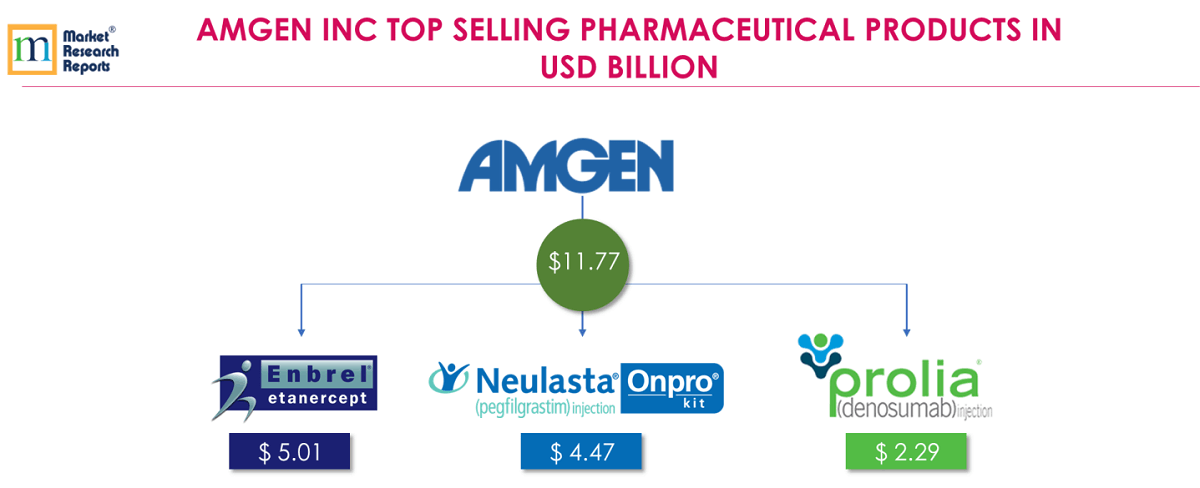
7 . 7.Amgen Inc:
Amgen focuses on innovative human therapeutics including Cardiovascular, Oncology/Hematology, Neuroscience, Inflammation and other areas. Amgen’s top 3 products for FY 2018 are Enbrel®, Neulasta® and Prolia®; contributes together USD 11.77 billion which is 52.31% of total Amgen’s revenue for FY 2018.
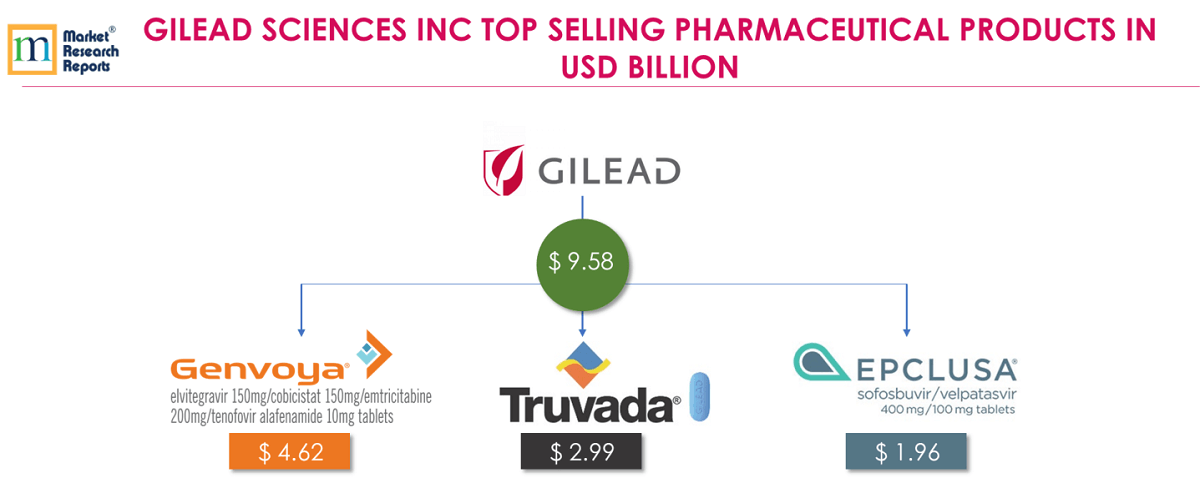
8 . 8.Gilead Sciences Inc:
Gilead’s primary areas of focus include HIV/AIDS, liver diseases, hematology/oncology and inflammation/respiratory diseases. Gilead’s top 3 products for FY 2018 are GENVOYA®, TRUVADA® and EPCLUSA®; contributes together USD 9.58 billion which is 43.29% of total Gilead’s revenue for FY 2018.
9 . 9.GlaxoSmithKline plc (GSK):
GSK's Pharmaceuticals business has a broad portfolio of innovative and established medicines, with leadership positions in respiratory and HIV. GSK’s top 3 products for FY 2018 are Triumeq®, Seretide®/ Advair® and Tivicay®; contributes together USD 8.92 billion which is 21.76% of total GSK's revenue for FY 2018.

10 . 10.Novartis AG:
Novartis has operates business in three segments – innovative medicines, Sandoz and Alcon. Novartis's top 3 products for FY 2018 are GILENYA™, COSENTYX® and LUCENTIS®; contributes together USD 8.22 billion which is 15.84% of total Novartis revenue for FY 2018.

COSENTYX® (Secukinumab) is approved for treatment of psoriasis, ankylosing spondylitis, and psoriatic arthritis. FY 2018 revenue from COSENTYX® was USD 2.83 billion.
LUCENTIS® (Ranibizumab) has been approved to treat the "wet" type of age-related macular degeneration (AMD, also ARMD), a common form of age-related vision loss. FY 2018 revenue from LUCENTIS® was USD 2.04 billion.
DISCLAIMER: Please be aware that, this analysis is meant for marketing and market research purpose only and should not be treated as medical advice.
Global Pharmaceuticals Industry Analysis and Trends 2023: Report covers growth forecasts for 39 countries along with 5 major geographic regions
Related Pharmaceutical Industry Articles
1. Global Pharmaceuticals Market Forecast: Drivers, Value Chain Analysis & Trends
2. Top 10 Oncology Pharma Companies in the World
3. Top 10 Antidiabetics Pharma Companies in the World
4. World’s Top 10 Pharmaceutical Companies by Revenue
5. U.S. Top 10 Pharmaceutical Companies by Market Turnover
6. Top 15 Pharma Companies in India
7. Top 6 Therapeutic Companies to Look out for Acute Renal Failure (Acute Kidney Injury)









